Moxonidine
| Clinical data | |
| Pronunciation | /мɒkˈsɒпɪдяп/ |
| Trade names | Physiotens |
| AHFS/Drugs.com | International names of drugs |
| Pregnancy category |
|
| Routes administration | Oral (tablets) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 88% (Tmax = 1 hour) |
| Protein binding | 7.2–10%[1][2] |
| Metabolism | Liver (10–20%)[2] |
| Metabolites | Dehydrogenated moxonidine (major), hydroxymethylmoxonidine, hydroxymoxonidine, dihydroxymoxonidine[3] |
| Elimination of half-life | ~2.2–2.8 hours |
| Excretion | Renal (90%),[4] feces (~1%)[2] |
| Identifiers | |
IUPAC name
| |
| Number of CAS |
|
| PubChem C.I.D. |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| CHAMBL |
|
| CompTox Control Panel (EPA) |
|
| ECHA InfoCard | 100.158.061 |
| Chemical and physical data | |
| Formula | C9H12ClN5O |
| Molar mass | 241.68 g mol−1 |
| 3D model (JSmol) |
|
Smiles
| |
InCHI
| |
| NY (what is this?) (check) | |
Moxonidine
(HOTEL) is a new generation alpha-2/imidazoline receptor agonist antihypertensive licensed drug for the treatment of mild to moderate essential hypertension.[5][6]
This may be important when thiazides, beta blockers, ACE inhibitors, and calcium channel blockers are not suitable or have failed to control blood pressure. In addition, it demonstrates a beneficial effect on parameters of insulin resistance syndrome, apparently independent of lowering blood pressure. It is also a growth hormone releaser.[7] Manufactured by Solvay Pharmaceuticals (acquired by Abbott in 2009) under the brand names Physiotens
and Moxon.
Composition and release form
The drug is sold in pharmacies in the form of small tablets for oral administration. Externally, they are white in color with a soluble thin shell. Packaged in blisters of 14 pieces and secondary cardboard packaging. One pack contains 1-2 cells of 14-28 tablets and includes instructions for use.
The active component of the drug is moxonidine. Its amount in 1 tablet is 200 mcg.
Additional substances: castor oil, cellulose, magnesium stearate, Tween 80, Klucel and Aerosil.
Cautions
| This section do not quote any sources . |
Moxonidine should be avoided in patients with moderate to severe renal impairment. You should also avoid abruptly stopping the drug. If it is necessary to discontinue concomitant treatment with beta-blockers, the beta blocker should be discontinued first, followed by moxonidine a few days later. Alcohol may enhance the hypotensive effect of moxonidine.
pharmachologic effect
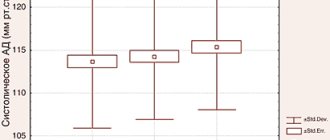
The drug belongs to the group of antihypertensive drugs. It is a direct agonist of imidazoline receptors. When used once, it helps to simultaneously restore the level of diastolic and systolic pressure. At the same time, it does not change the activity of the heart and does not affect the heart rate.
The drug is highly digestible, regardless of the time of meal. The biological availability of the drug is at least 88%. The highest concentration of the drug in the red channel is observed after 30 minutes or a maximum of 3 hours.
Moxonidine
Suction
After oral administration, moxonidine is rapidly and almost completely absorbed from the upper gastrointestinal tract. Absolute bioavailability is approximately 88%, indicating no significant first-pass effect. The time to reach maximum concentration is about 1 hour. Food intake does not affect the pharmacokinetics of the drug.
Distribution
The binding to plasma proteins is 7.2%.
Metabolism
The main metabolite is dehydrogenated moxonidine. The pharmacodynamic activity of dehydrogenated moxonidine is about 10% compared to moxonidine.
Removal
The half-life (T1/2) of moxonidine and metabolites is 2.5 and 5 hours, respectively. Within 24 hours, over 90% of moxonidine is excreted by the kidneys (about 78% unchanged and 13% as dehydriromoxonidine, other metabolites in the urine do not exceed 8% of the dose taken). Less than 1% is excreted through the intestines.
Pharmacokinetics in patients with arterial hypertension
Compared with healthy volunteers, patients with arterial hypertension show no changes in the pharmacokinetics of moxonidine.
Pharmacokinetics in old age
Clinically insignificant changes in the pharmacokinetic parameters of moxonidine were noted in elderly patients, probably due to a decrease in the intensity of its metabolism and/or slightly higher bioavailability.
Pharmacokinetics in children
Moxonidine is not recommended for use in patients under 18 years of age, and therefore pharmacokinetic studies have not been conducted in this group.
Pharmacokinetics in renal failure
Moxonidine excretion is significantly correlated with creatinine clearance (CC). In patients with moderate renal failure (creatinine clearance in the range of 30-60 ml/min), steady-state plasma concentrations and final T1/2 are approximately 2 and 1.5 times higher than in individuals with normal renal function (creatinine clearance more than 90 ml/min). min).
In patients with severe renal failure (creatinine clearance less than 30 ml/min), steady-state plasma concentrations and final T1/2 are 3 times higher than in patients with normal renal function. The administration of multiple doses of moxonidine leads to predictable accumulation in the body of patients with moderate and severe renal failure.
In patients with end-stage renal failure (creatinine clearance less than 10 ml/min) on hemodialysis, steady-state plasma concentrations and final T1/2 are 6 and 4 times higher, respectively, than in patients with normal renal function.
In patients with moderate renal failure, the maximum concentration of moxonidine in the blood plasma is 1.5-2 times higher.
In patients with impaired renal function, the dosage should be adjusted individually.
Moxonidine is excreted to a small extent during hemodialysis.
Contraindications
It is prohibited to take the medicine for the following conditions:
- Individual intolerance to the active or auxiliary components.
- Acute form of bradycardia.
- Severe arrhythmia.
- Glaucoma.
- Mental disorders.
- Cardiac failure.
- Previous Quincke's edema.
- The period of bearing a child and breastfeeding.
- Children under 18 years of age.
- Disturbance of the liver and kidneys.
- Angina pectoris.
- Lactose deficiency or intolerance.
- Various pathologies of the circulatory system.
Synergistic effects of drugs
| This section do not quote any sources . |
Concomitant use of moxonidine and thiazide diuretics such as hydrochlorothiazide gave a synergistic antihypertensive effect.[9]
Side effects
As a result of treatment with Moxonidine, patients may experience the following adverse reactions:
- High fatigue, malaise.
- Migraine.
- Sleep disorder - insomnia or drowsiness.
- Dizziness, headache.
- Dyspeptic manifestations - dry mouth, discomfort in the epigastrium, nausea, vomiting, abnormal stool.
- Increased nervousness and irritability.
- Swelling of peripheral tissues.
- A sharp decrease in pulse and blood pressure.
- Allergic signs are skin rash and severe itching.
Safety pharmacology
| This section do not quote any sources . |
Routine toxicological studies have not provided evidence that moxonidine has any teratogenic, mutagenic or carcinogenic potential. There was no evidence of serious adverse effects on organs or organ systems, and the drug has not been shown to have harmful effects on perinatal or postnatal growth and development.
Drug overdose
If medical recommendations are not followed and the maximum volume of the drug is exceeded, an overdose may develop. The condition is determined by severe symptoms:
- Intense headache and dizziness.
- Nausea followed by vomiting.
- Painful sensations in the stomach area.
- Weakness and severe malaise.
- Extensive reduction in pressure.
- Dryness in the mouth.
If the above symptoms occur, you should definitely consult a doctor. Treatment involves relief of symptoms. Prescribed medication, gastric lavage, parenteral administration of saline. To suppress the symptoms of bradycardia, the use of Atropine is indicated.
Mechanism of action
Moxonidine is a selective agonist for imidazoline receptor subtype 1 (I1).[5] This receptor subtype is found in both the rostral ventrolateral pressor and ventromedial depressor areas of the medulla oblongata. Thus, moxonidine causes a decrease in sympathetic nervous system activity and, as a consequence, a decrease in blood pressure.
Compared to older centrally acting antihypertensive agents, moxonidine binds to the imidazoline I.1 receptor rather than the α2 receptor. In contrast, clonidine binds to both receptors with almost equal affinity. Moxonidine has an affinity for I1 that is 33 times greater than α2, compared to clonidine, which is only four times greater.[8]
In addition, moxonidine may also promote sodium excretion, improve insulin resistance and glucose tolerance, and protect against end organ damage in hypertension such as kidney disease and cardiac hypertrophy.
special instructions
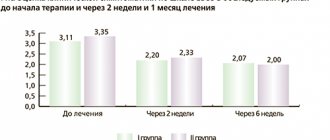
The use of an antihypertensive drug for therapeutic purposes requires constant monitoring of blood pressure, heart function and heart rate.
During the course of treatment, you should completely avoid alcoholic beverages. It is recommended not to drive vehicles or engage in activities that require concentration, memory and high mental activity.
You need to stop taking the drug gradually, gradually reducing the dose until complete withdrawal.
If the use of Moxonidine was combined with beta-blockers and it is necessary to discontinue both drugs, the latter are removed first. After some time, the antihypertensive drug is discontinued.
Pharmacodynamic properties
| This section do not quote any sources . |
Effect on insulin resistance
In all animal models of insulin resistance, moxonidine had a striking effect on the development of insulin resistance, hyperinsulinemia, and impaired glucose homeostasis. Given the importance of insulin resistance as a risk factor for cardiovascular disease, it is significant that it has been shown to improve insulin sensitivity.
Moxonidine-SZ
Suction
After oral administration, moxonidine is rapidly and almost completely absorbed from the upper gastrointestinal tract. Absolute bioavailability is approximately 88%. The time to reach maximum concentration is about 1 hour. Food intake does not affect the pharmacokinetics of the drug.
Distribution
The binding to plasma proteins is 7.2%.
Metabolism
The main metabolite is dehydrogenated moxonidine. The pharmacodynamic activity of dehydrogenated moxonidine is about 10% compared to moxonidine.
Removal
The half-life (T1/2) of moxonidine and metabolite is 2.5 and 5 hours, respectively. Within 24 hours, over 90% of moxonidine is excreted by the kidneys (about 78% unchanged and 13% as dehydriroxonidine, other metabolites in the urine do not exceed 8% of the dose taken). Less than 1% of the dose is excreted through the intestines.
Pharmacokinetics in
patients with arterial hypertension
Compared with healthy volunteers, patients with arterial hypertension do not show changes in the pharmacokinetics of moxonidine.
Pharmacokinetics in old age
Clinically insignificant changes in the pharmacokinetic parameters of moxonidine were noted in elderly patients, probably due to a decrease in the intensity of its metabolism and/or slightly higher bioavailability.
Pharmacokinetics in children
Moxonidine is not recommended for use in patients under 18 years of age, and therefore pharmacokinetic studies have not been conducted in this group.
Pharmacokinetics in renal failure
Moxonidine excretion is significantly correlated with creatinine clearance (CC). In patients with moderate renal failure (creatinine clearance in the range of 30-60 ml/min), steady-state plasma concentrations and final Ti/2 are approximately 2 and 1.5 times higher than in patients with normal renal function (creatinine clearance more than 90 ml/min). min.).
In patients with severe renal failure (creatinine clearance less than 30 ml/min.), steady-state plasma concentrations and final T1/2 are 3 times higher than in patients with normal renal function. Prescription of multiple doses of moxonidine leads to predictable accumulation in the body
patients with moderate to severe renal failure. In patients with end-stage renal failure (creatinine clearance less than 10 ml/min) on hemodialysis, steady-state plasma concentrations and final T1/2 are 6 and 4 times higher, respectively, than in patients with normal renal function. In all groups, the maximum concentration of moxonidine in blood plasma was 1.5-2 times higher. In patients with impaired renal function, the dosage should be adjusted individually.
Moxonidine is excreted to a small extent during hemodialysis.

![Table 2. Hemodynamic definition of PH (adapted from [2] with modifications)](https://ms-pi.ru/wp-content/uploads/tablica-2-gemodinamicheskoe-opredelenie-lg-adaptirovano-iz-2-s-izmeneniyami-330x140.jpg)