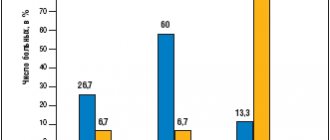
Effect of risperidone
The mechanism of action is caused by the effect on dopamine, serotonin, adrenergic and histamine receptors of nerve cells. Due to the fact that in many endogenous, organic mental diseases the functions of these receptor systems are changed, risperidone has a fairly broad therapeutic effect:
- For productive symptoms of psychosis and schizophrenia (delusions and hallucinations), for negative ones - apathy, isolation, lack of emotions, autism.
- Reduces pathologically elevated mood (mania).
- Calming, anti-anxiety, hypnotic effect.
- Reduces (or removes) aggressiveness, self-aggression.
- Anti-suicidal action.
- Reducing the pathological craving for drugs and alcohol in case of addiction.
- Removes obsessive thoughts, actions and sensations.
Risperidone (Rispolept, Torendo, Rispaxol)
Contraindications
Hypersensitivity to risperidone.
Dosage
Individual. When taken orally, the initial dose for adults is 0.25-2 mg/day, on the 2nd day - 4 mg/day. Then the dose can either be kept at the same level or, if necessary, adjusted. Typically, the optimal therapeutic dose, depending on the indications, is in the range of 0.5-6 mg/day. In some cases, a slower dose escalation and lower initial and maintenance doses may be justified.
For schizophrenia, for elderly patients, as well as for concomitant liver and kidney diseases, an initial dose of 500 mcg 2 times a day is recommended. If necessary, the dose can be increased to 1-2 mg 2 times a day.
Maximum dose: When using risperidone at a dose of more than 10 mg/day, there is no increase in effectiveness compared to lower doses, but the risk of developing extrapyramidal symptoms increases. The safety of risperidone in doses greater than 16 mg/day has not been studied, so further excess of the dose is not allowed.
Side effects
From the side of the central nervous system: often - insomnia, agitation, anxiety, headache; possible - drowsiness, fatigue, dizziness, impaired ability to concentrate, impaired vision; rarely - extrapyramidal symptoms (including tremor, rigidity, hypersalivation, bradykinesia, akathisia, acute dystonia). Patients with schizophrenia may experience tardive dyskinesia, NMS, thermoregulation disorders, and seizures.
From the digestive system: constipation, dyspeptic symptoms, nausea, vomiting, abdominal pain, increased activity of liver enzymes.
From the reproductive system: priapism, erectile dysfunction, ejaculation disorders, orgasm disorders.
From the cardiovascular system: rarely - orthostatic hypotension and reflex tachycardia, arterial hypertension.
From the endocrine system: galactorrhea, gynecomastia, menstrual irregularities, amenorrhea, weight gain.
From the hematopoietic system: a slight decrease in the number of neutrophils and/or platelets.
Allergic reactions: rhinitis, skin rash, angioedema.
Other: urinary incontinence.
Drug interactions
With the simultaneous use of inducers of microsomal liver enzymes, a decrease in the concentration of risperidone in the blood plasma is possible.
When used simultaneously with phenothiazine derivatives, tricyclic antidepressants and beta-blockers, the concentration of risperidone in the blood plasma may increase.
When used simultaneously with carbamazepine, the concentration of risperidone in the blood plasma is significantly reduced.
When used concomitantly, risperidone reduces the effects of levodopa and other dopamine receptor agonists.
When used simultaneously with fluoxetine, an increase in the concentration of risperidone in the blood plasma is possible.
special instructions
Use with caution in patients with diseases of the cardiovascular system (including heart failure, myocardial infarction, cardiac muscle conduction disorders), as well as in cases of dehydration, hypovolemia or cerebrovascular disorders. In this category of patients, the dose should be increased gradually.
The risk of developing orthostatic hypotension is especially increased in the initial period of dose selection. If hypotension occurs, dose reduction should be considered.
When using drugs that have the properties of dopamine receptor antagonists, the occurrence of tardive dyskinesia, characterized by involuntary rhythmic movements (mainly of the tongue and/or face), was noted. There are reports that the occurrence of extrapyramidal symptoms is a risk factor for the development of tardive dyskinesia. Risperidone causes extrapyramidal symptoms to a lesser extent than classical antipsychotics. If symptoms of tardive dyskinesia occur, discontinuation of all antipsychotic medications should be considered.
If NMS develops, all antipsychotics, including risperidone, should be discontinued.
Risperidone should be used with caution in patients with Parkinson's disease, as it is theoretically possible that the disease may worsen.
Classical antipsychotics are known to lower the seizure threshold. Given this, risperidone is recommended to be used with caution in patients with epilepsy.
Risperidone should be used with caution in combination with other centrally acting drugs.
When discontinuing carbamazepine and other hepatic enzyme inducers, the dose of risperidone should be reconsidered and, if necessary, reduced.
During the treatment period, patients should be advised to refrain from overeating due to the possibility of weight gain.
There are no data on the safety of risperidone in children under 15 years of age.
Impact on the ability to drive vehicles and operate machinery
During the treatment period, until individual sensitivity to risperidone is determined, patients should avoid driving vehicles and other activities that require high concentration and speed of psychomotor reactions.
Pregnancy and lactation
Use during pregnancy is possible if the expected benefit of therapy for the mother outweighs the potential risk to the fetus. If use is necessary during lactation, breastfeeding should be discontinued.
Use in childhood
There are no data on the safety of risperidone in children under 15 years of age.
For impaired renal function
For schizophrenia with concomitant kidney diseases, an initial dose of 500 mcg 2 times a day is recommended. If necessary, the dose can be increased to 1-2 mg 2 times a day.
For liver dysfunction
For schizophrenia with concomitant liver diseases, an initial dose of 500 mcg 2 times a day is recommended. If necessary, the dose can be increased to 1-2 mg 2 times / day.
Use in old age
For schizophrenia, an initial dose of 500 mcg 2 times a day is recommended for elderly patients. If necessary, the dose can be increased to 1-2 mg 2 times a day.
Release form
One of the advantages of this drug is the presence of all the dosage forms necessary for psychiatrists: tablets, drops, extended-release injections (depot).
Pills
2 or 4 milligrams per package of 10,20,30,50,60 or 100 pieces
Drops
. In bottles of 30 or 100 milliliters. A special graduated pipette is included, which is used to determine the amount of a single dose to take.
Depot
. In powder form for the preparation of a long-acting intramuscular suspension. Dosages: 25, 37.5 and 50 milligrams. It is used as an intramuscular injection, which will ensure slow release and entry of the drug into the blood over 2 - 3 weeks. Produced under the name RISPOLEPT CONST.
Rispolept oral solution 1 mg/ml 30 ml bottle in Moscow
Inside,
regardless of food intake.
Schizophrenia
Adults.
Rispolept® can be prescribed 1 or 2 times a day. The initial dose of Rispolept® is 2 mg/day. On the second day, the dose should be increased to 4 mg/day. From this point on, the dose can either be kept at the same level or individually adjusted if necessary. Typically the optimal dose is 4–6 mg/day. In some cases, a slower dose escalation and lower initial and maintenance doses may be justified.
Doses above 10 mg/day do not show greater efficacy than lower doses and may cause extrapyramidal symptoms. Due to the fact that the safety of doses above 16 mg/day has not been studied, doses above this level cannot be used.
Benzodiazepines can be added to Rispolept® therapy if additional sedation is required.
Elderly patients.
The recommended initial dose is 0.5 mg per dose 2 times a day. The dose can be individually increased by 0.5 mg 2 times a day to 1–2 mg 2 times a day.
Teenagers over 13 years old.
The recommended initial dose is 0.5 mg once daily in the morning or evening. If necessary, the dose can be increased after at least 24 hours by 0.5–1 mg per day to the recommended dose of 3 mg/day if well tolerated. The safety of doses above 6 mg/day has not been studied.
For patients who experience persistent drowsiness, it is recommended to take half the daily dose 2 times a day.
There is no information on the use of the drug for the treatment of schizophrenia in children under 13 years of age.
Behavioral disorders in patients with dementia
The recommended initial dose is 0.25 mg per dose 2 times a day. If necessary, the dose can be increased individually by 0.25 mg 2 times a day, no more than every other day. For most patients, the optimal dose is 0.5 mg 2 times a day. However, some patients are advised to take 1 mg 2 times a day.
Once the optimal dose is reached, it may be recommended to take the drug once a day.
Bipolar disorders with mania
Adults.
The recommended initial dose of the drug is 2 or 3 mg/day at a time. If necessary, this dose can be increased at least after 24 hours by 1 mg/day. For most patients, the optimal dose is 1–6 mg/day.
Teenagers and children over 10 years old.
The recommended initial dose is 0.5 mg once daily in the morning or evening. If necessary, the dose can be increased after at least 24 hours by 0.5–1 mg/day to the recommended dose of 2.5 mg/day if well tolerated. For most patients, the optimal dose is 0.5–6 mg/day.
The safety of doses above 6 mg/day has not been studied.
For patients who experience persistent drowsiness, it is recommended to take half the daily dose 2 times a day.
There is no information on the use of the drug for the treatment of bipolar disorders in children under 10 years of age.
Behavioral disorders
Patients weighing 50 kg or more.
The recommended initial dose of the drug is 0.5 mg once a day. If necessary, this dose can be increased by 0.5 mg/day no more than every other day. For most patients, the optimal dose is 1 mg/day. However, for some patients, 0.5 mg/day is preferable, while some require an increase in dose to 1.5 mg/day.
Patients weighing less than 50 kg.
The recommended initial dose of the drug is 0.25 mg once a day. If necessary, this dose can be increased by 0.25 mg/day no more than every other day. For most patients, the optimal dose is 0.5 mg/day. However, for some patients, 0.25 mg/day is preferable, while some require an increase in dose to 0.75 mg/day.
Long-term use of the drug Rispolept® in adolescents should be carried out under the constant supervision of a physician.
Use in children under 5 years of age has not been studied.
Autism in children and adolescents
The dose of Rispolept® should be selected individually. The recommended initial dose of the drug is 0.25 mg/day for patients weighing less than 20 kg and 0.5 mg/day for patients weighing 20 kg or more. On the 4th day of administration, the dose can be increased by 0.25 mg/day for patients weighing less than 20 kg and by 0.5 mg/day per day for patients weighing 20 kg or more.
This dose should be used until approximately the 14th day of treatment, when it is necessary to evaluate the effectiveness. Further increase in dose is carried out only in case of lack of effectiveness. The dose can be increased at intervals of 2 weeks or more by 0.25 mg/day for patients weighing less than 20 kg and by 0.5 mg/day for patients weighing 20 kg or more.
In clinical studies, the maximum daily dose did not exceed 1.5 mg/day for patients weighing less than 20 kg and 2.5 mg/day for patients weighing 20 kg or more, and 3.5 mg/day for patients with weight body more than 45 kg.
Table
Doses of the drug Rispolept® in the treatment of autism in children (per day)
| Body weight, kg | Days 1–3, mg | Days 4–14+, mg | Increase the dose (if necessary) | Recommended dose, mg |
| Less than 20 | 0,25 | 0,5 | +0.25 mg after 2 weeks or more | 0,5–1,5 |
| 20 or more | 0,5 | 1 | +0.5 mg after 2 weeks or more | 1–2,5* |
* For patients weighing more than 45 kg, higher doses may be required, the maximum dose studied is 3.5 mg/day.
Rispolept® can be prescribed 1 or 2 times a day.
For patients who experience persistent drowsiness, it is recommended to take a daily dose 1 time before bedtime or 2 times a day.
If consistent efficacy is observed, the decision may be made to gradually reduce the dose to achieve an optimal balance of efficacy and safety.
Use in children under 5 years of age has not been studied.
Other patient groups
Patients with liver and kidney diseases.
Patients with kidney disease have a reduced ability to eliminate the active antipsychotic fraction compared to other patients. In patients with liver disease, there is an increased concentration of the free fraction of risperidone in the blood plasma.
The initial and maintenance dose, in accordance with the indications, should be reduced by 2 times; the dose increase in patients with liver and kidney diseases should be carried out more slowly.
Rispolept® should be prescribed with caution in this category of patients.
Indications
Risperidone was originally used to treat schizophrenia and psychosis. But as experience has accumulated about the action of this medicine, the indications for its use have expanded. Currently indicated for the following diseases and conditions:
- Schizophrenia
- Schizotypal disorder
- Organic mental disorders
- Schizoid personality disorder
- Manic and hypomanic states in bipolar affective disorder, schizoaffective disorder
- Psychoses
- Delusions: paranoia, paranoid, paraphrenia
- Depressive and anxiety states, neuroses and neurosis-like conditions
- Aggressiveness
- Autism
The drug is recommended for adults and children over 13 years of age. At a younger age, it is prescribed by the doctor’s decision in cases where the potential risk of side and negative effects of the drug is less than the risk of worsening the condition as the disease progresses.
Instructions for use RISPOLEPT®
The drug should be used with caution in patients with diseases of the cardiovascular system (including chronic heart failure, previous myocardial infarction, conduction disorders), dehydration, hypovolemia, cerebrovascular accident, Parkinson's disease, convulsions (including history), severe renal or hepatic failure, drug abuse or drug dependence, conditions predisposing to the development of torsade de pointes (bradycardia, electrolyte imbalance, concomitant use of drugs that prolong the QT interval), brain tumors , intestinal obstruction, in cases of acute drug overdose, in Reye's syndrome (since the antiemetic effect of risperidone can mask the symptoms of these conditions), during pregnancy, in patients under the age of 15 years (since the safety and effectiveness of the drug have not been established) .
In case of schizophrenia, at the beginning of treatment with Rispolept, it is recommended to gradually discontinue previous therapy if clinically justified. At the same time, if patients are transferred from depot therapy with antipsychotic drugs, it is recommended to start Rispolept therapy instead of the next scheduled injection. The need to continue current antiparkinsonian drug therapy should be periodically assessed.
Due to the alpha-adrenergic blocking effect of Rispolept, orthostatic hypotension may occur, especially during the initial dose selection period. If arterial hypotension occurs, dose reduction should be considered. In patients with diseases of the cardiovascular system, as well as in cases of dehydration, hypovolemia or cerebrovascular disorders, the dose should be increased gradually.
There are reports that the occurrence of extrapyramidal symptoms is a risk factor for the development of tardive dyskinesia. Rispolept is less likely to cause extrapyramidal symptoms than classic antipsychotics. If signs and symptoms of tardive dyskinesia occur, discontinuation of all antipsychotic medications should be considered.
In case of development of NMS, characterized by hyperthermia, muscle rigidity, instability of autonomic functions, disturbances of consciousness and increased CPK levels, it is necessary to discontinue all antipsychotic drugs, including Rispolept.
When carbamazepine and other liver enzyme inducers are discontinued, the dose of Rispolept should be reduced.
During treatment with Rispolept, patients should be advised to refrain from overeating due to the possibility of weight gain.
Use in pediatrics
Data on the safety and effectiveness of Rispolept in children under 15 years of age
are absent, so prescribing the drug to this category of patients is not recommended. Long-term use of Rispolept in adolescents should be carried out under the constant supervision of a physician.
Impact on the ability to drive vehicles and operate machinery
During the treatment period, until individual sensitivity to the drug is determined, patients should avoid driving vehicles and other activities that require high concentration and speed of psychomotor reactions.
Use during pregnancy and breastfeeding
There are no absolute contraindications to taking risperidone during pregnancy and lactation. However, due to the lack of special research in this direction, use during pregnancy is allowed only in cases described by the following formulation: when the benefits of taking it for the pregnant woman outweigh the potential risks to the fetus.
Due to the fact that risperidone passes into breast milk, it is recommended to transfer the child to artificial nutrition for the period of treatment of a breastfeeding mother.
Rispolept drops 1mg/ml 30ml N1 (Johnson)
Use in elderly patients with dementia
Increased mortality in older patients with dementia
Elderly patients with dementia treated with atypical antipsychotics experienced increased mortality compared with placebo in studies of atypical antipsychotics, including risperidone. When using risperidone in this population, the incidence of death was 4.0% for patients taking risperidone compared with 3.1% for placebo. The mean age of patients who died was 86 years (range, 67–100 years). Data collected from two large observational studies show that older patients with dementia treated with typical antipsychotic medications also have a slightly increased risk of death compared with patients not treated. At present, insufficient data have been collected to accurately assess this risk. The reason for the increase in this risk is also unknown. Also unknown is the extent to which the increased mortality may be attributable to antipsychotic drugs rather than to the characteristics of this patient population.
Combined use with furosemide
In elderly patients with dementia, there was an increased mortality rate when taking furosemide and oral risperidone concomitantly (7.3%, mean age 89 years, range 75-97 years) compared with the risperidone alone group (3.1%, mean age 84 years , range 70-96 years) and the furosemide-only group (4.1%, mean age 80 years, range 67-90 years). An increase in mortality in patients taking risperidone with furosemide was observed in 2 of 4 clinical studies. Concomitant use of risperidone with other diuretics (mainly low-dose thiazide diuretics) was not associated with an increase in mortality.
No pathophysiological mechanisms have been established to explain this observation. However, special care should be taken when prescribing the drug in such cases. Before prescribing, the risk/benefit ratio must be carefully assessed. There was no increase in mortality in patients taking other diuretics concomitantly with risperidone. Regardless of treatment, dehydration is a common risk factor for mortality and should be carefully monitored in older patients with dementia.
In elderly patients with dementia, an increase in cerebrovascular adverse events (acute and transient cerebrovascular accidents), including patient deaths (mean age 85 years, range 73-97 years) was observed with risperidone compared with placebo.
Cardiovascular effects
In placebo-controlled clinical trials, an approximately 3-fold increased risk of cerebrovascular side effects was observed in patients with dementia taking certain atypical antipsychotic drugs. Pooled data from 6 placebo-controlled studies involving primarily elderly patients with dementia (age >65 years) demonstrate that cerebrovascular adverse events (serious and non-serious) occurred in 3.3% (33/1009) of patients treated with risperidone. and in 1.2% (8/712) of patients receiving placebo. The risk ratio was 2.96 (1.34, 7.50) with a 95% confidence interval. The mechanism by which this risk increases is unknown. An increased risk cannot be excluded for other antipsychotic drugs, as well as for other patient populations. Rispolept® should be used with caution in patients with risk factors for stroke.
The risk of cerebrovascular side effects is much higher in patients with mixed or vascular dementia compared to patients with Alzheimer's dementia. Therefore, patients with any type of dementia other than Alzheimer's should not take risperidone.
Physicians should assess the risk/benefit ratio of using Rispolept® in elderly patients with dementia, taking into account the precursors of stroke risk individually for each patient.
Patients and caregivers should be cautioned to immediately report signs and symptoms of cardiovascular events, such as sudden weakness or stiffness/numbness in the face, legs, arms, as well as difficulty speaking and vision problems. All possible treatment options should be considered, including discontinuation of risperidone.
Rispolept® should only be used for the short-term treatment of persistent aggression in patients with moderate to severe Alzheimer's dementia, as an adjunct to non-pharmacological treatments when they are ineffective or of limited effectiveness, and when there is a risk of harm to the patient himself or herself to other persons.
Patients' condition and the need for continued risperidone therapy should be continually assessed.
Orthostatic hypotension
Risperidone has alpha-blocking activity and may therefore cause orthostatic hypotension in some patients, especially during initial dose titration. Clinically significant hypotension has been observed in the post-marketing period when used concomitantly with antihypertensive drugs. Rispolept® should be used with caution in patients with known cardiovascular disease (eg, heart failure, myocardial infarction, cardiac conduction disorders, dehydration, hypovolemia or cerebrovascular disease). Appropriate dose adjustment is also necessary. It is recommended to evaluate the possibility of dose reduction if hypotension occurs.
Tardive dyskinesia and extrapyramidal disorders
Drugs with dopamine receptor antagonist properties can cause tardive dyskinesia, which is characterized by rhythmic involuntary movements, mainly of the tongue and/or facial muscles. The occurrence of extrapyramidal symptoms is a risk factor for the development of tardive dyskinesia. If a patient experiences objective or subjective symptoms indicating tardive dyskinesia, the advisability of discontinuing all antipsychotic drugs, including Rispolept®, oral solution, should be considered.
Neuroleptic malignant syndrome (NMS)
Antipsychotics, including risperidone, may cause neuroleptic malignant syndrome (NMS), which is characterized by hyperthermia, muscle rigidity, instability of autonomic nervous system function, depression of consciousness, and increased serum concentrations of creatine phosphokinase. Myoglobinuria (rhabdomyolysis) and acute renal failure may also occur in patients with NMS. If a patient experiences objective or subjective symptoms of NMS, all antipsychotic drugs, including Rispolept®, must be immediately discontinued.
Parkinson's disease and dementia with Lewy bodies
Antipsychotic medications, including Rispolept®, should be prescribed with caution to patients with Parkinson's disease or dementia with Lewy bodies. Both groups of patients have an increased risk of developing neuroleptic malignant syndrome and increased sensitivity to antipsychotic drugs (including dullness of pain sensitivity, confusion, postural instability with frequent falls and extrapyramidal symptoms). Parkinson's disease may worsen when taking risperidone.
Hyperglycemia and diabetes mellitus
Hyperglycemia, diabetes mellitus, and exacerbation of existing diabetes mellitus were observed during treatment with Rispolept®. It is likely that weight gain prior to treatment is also a predisposing factor. Very rarely, ketoacidosis and rarely, diabetic coma can occur. All patients should be clinically monitored for symptoms of hyperglycemia (such as polydipsia, polyuria, polyphagia and weakness). Patients with diabetes mellitus should be regularly monitored for worsening glucose control.
Weight gain
During treatment with Rispolept®, a significant increase in body weight was observed. It is necessary to monitor patients' body weight.
Hyperprolactinemia
Based on the results of tissue culture studies, it has been suggested that the growth of breast tumor cells may be stimulated by prolactin. Although clinical and epidemiological studies have not shown a clear association between hyperprolactinemia and antipsychotic drug use, caution should be exercised when prescribing risperidone to patients with a history of this. Rispolept® should be used with caution in patients with existing hyperprolactinemia and in patients with possible prolactin-dependent tumors.
Prolongation of the QT interval
QT prolongation has been observed very rarely during post-marketing surveillance. As with other antipsychotics, caution should be exercised when prescribing Rispolept® to patients with known cardiovascular diseases, family history of QT interval prolongation, bradycardia, electrolyte imbalance (hypokalemia, hypomagnesemia), as this may increase the risk of an arrhythmogenic effect; and when used together with drugs that prolong the QT interval.
Convulsions
Rispolept® should be used with caution in patients with a history of seizures or other medical conditions that may lower the seizure threshold.
Priapism
Priapism may occur with risperidone due to alpha-blocking effects.
Body temperature regulation
Antipsychotic drugs are associated with such undesirable effects as disruption of the body's ability to regulate temperature. Caution should be exercised when prescribing Rispolept® to patients with conditions that may contribute to an increase in core body temperature, such as intense physical activity, dehydration, exposure to high external temperatures, or concomitant use of drugs with anticholinergic activity.
Venous thromboembolism
Cases of venous thromboembolism have been reported with the use of antipsychotic drugs. Since patients taking antipsychotic drugs are often at risk of developing venous thromboembolism, all possible risk factors should be identified before and during treatment with Rispolept®, and preventive measures should be taken.
Children and teenagers
Before prescribing Rispolept® to children or adolescents with mental retardation, their condition must be carefully assessed for the presence of physical or social causes of aggressive behavior, such as pain or inadequate demands of the social environment.
The sedative effect of risperidone should be carefully monitored in this population due to the possible effect on learning ability. Changing the timing of risperidone administration may improve control of the effects of sedation on attention in adolescents and children. Risperidone use was associated with mean increases in body weight and body mass index. Height changes in longitudinal studies were within expected age-related norms. The effects of long-term use of risperidone on sexual development and growth have not been fully studied.
Due to the possible impact of prolonged hyperprolactinemia on growth and sexual development in children and adolescents, regular clinical assessment of hormonal status should be carried out, including measurement of height, weight, monitoring of sexual development, menstrual cycle and other possible prolactin-dependent effects.
During treatment with risperidone, regular monitoring for the presence of extrapyramidal symptoms and other movement disorders should be carried out.
Side effects
The drug is usually well tolerated. According to clinic doctors, it is one of the well-tolerated antipsychotics. In approximately 10% of cases, the following side effects are possible.
- Increased muscle tone, restlessness, trembling, feeling of stiffness, “extrapyramidal syndrome,” parkinsonism. To prevent these phenomena, correctors may be additionally prescribed: trihexyphenidyl (Cyclodol), biperiden (Akineton, Mendylex, Bezac), amantadine (PK-Merz), etc.
- Headache.
- Insomnia.
Rare but unpleasant side effects include metabolic (metabolic) disorders: weight gain and the development of diabetes. As a rule, such effects develop in persons predisposed to this.
Therefore, to avoid them, it is recommended to conduct an examination with blood tests before starting therapy to exclude an increase in glucose and prolactin in the blood.
Rispolept®
Side effects of the drug Rispolept® in therapeutic doses are given with a distribution by frequency and system-organ classes. Determination of the frequency of side effects: very often (≥1/10), often (≥1/100 and <1/10), infrequently (≥1/1000 and <1/100), rarely (≥1/10,000 and <1 /1000), very rare (< 1/10,000).
Infections:
very often - in elderly patients with dementia - urinary tract infections; often - nasopharyngitis, upper respiratory tract infections, sinusitis, urinary tract infections, in elderly patients with dementia - pneumonia, phlegmon, in children - rhinitis, influenza-like diseases; uncommon - ear infections, viral infections, pharyngitis, tonsillitis, bronchitis, eye infections, localized infections, cystitis, onychomycosis, acrodermatitis, bronchopneumonia, respiratory tract infections, tracheobronchitis.
Mental disorders:
very often - insomnia; often - anxiety, nervousness, in elderly patients with dementia - confusion, in children - lethargy; infrequently - agitation, flattening of affect, sleep disturbances, weakened libido, anorgasmia; very rarely - mania.
From the nervous system:
very often - parkinsonism (including extrapyramidal disorders, cogwheel syndrome, akinesia, bradykinesia, hypokinesia, muscle rigidity), in children - drowsiness, headaches, sedation; often - akathisia (including restlessness), drowsiness, dizziness, sedation, tremor, dystonia (including muscle spasms, involuntary muscle contractions, muscle contracture, involuntary eye movements, tongue paralysis), lethargy, postural dizziness, dyskinesia (including muscle twitching, chorea and choreoathetosis), fainting, in elderly patients with dementia - a depressed state, in children - dysarthria, impaired attention, gait disturbance; uncommon - lack of response to stimuli, loss of coordination, loss of consciousness, speech impairment, hypoesthesia, movement disorder, tardive dyskinesia, cerebral ischemia, cerebrovascular disorders, NMS.
From the senses:
often - impaired visual acuity, ear pain, in elderly patients with dementia - conjunctivitis; uncommon - tinnitus, conjunctival hyperemia, blurred vision, involuntary rotation of the eyeballs, swelling of the eyelids, periorbital edema, crust formation on the edges of the eyelids, dry eyes, increased lacrimation, photophobia, increased intraocular pressure.
From the cardiovascular system:
often - tachycardia, orthostatic hypotension, decreased blood pressure, in elderly patients with dementia - transient ischemic attack, myocardial infarction, stroke, in children - palpitations; uncommon - sinus bradycardia, sinus tachycardia, palpitations, AV block, right and left bundle branch block, flushing; very rarely - atrial fibrillation.
From the respiratory system:
often - nasal congestion, shortness of breath, nosebleeds, sinus congestion, in elderly patients with dementia - cough, rhinorrhea, in children - cough, rhinorrhea, pain in the larynx and pharynx, pulmonary congestion; uncommon - wheezing, aspiration pneumonia, dysphonia, productive cough, airway obstruction, moist rales, respiratory distress, nasal swelling, hyperventilation; very rarely - sleep apnea syndrome.
From the digestive system:
often - nausea, constipation, dyspepsia, vomiting, diarrhea, salivation, dry mouth, stomach discomfort, abdominal pain; in elderly patients with dementia - dysphagia, fecaloma; infrequently - encopresis, gastritis, swelling of the lips, cheilitis, aptialism; very rarely - intestinal obstruction, pancreatitis, jaundice.
From the hematopoietic system:
often - anemia; uncommon - granulocytopenia; very rarely - thrombocytopenia, agranulocytosis.
Allergic reactions:
uncommon - hypersensitivity; very rarely - Quincke's edema, anaphylactic shock.
From the endocrine system:
uncommon - hyperprolactinemia, diabetic coma; very rarely - impaired ADH secretion.
From the side of metabolism:
often in elderly patients with dementia there is a decrease in appetite, in children there is an increase in appetite; infrequently - polydipsia, anorexia; rarely - diabetic ketoacidosis, diabetes mellitus, hypoglycemia, water intoxication.
Dermatological reactions:
often - rash, dry skin, dandruff, seborrheic dermatitis, hyperkeratosis, in elderly patients with dementia - erythema, in children - itching, acne; uncommon - skin pigmentation disorders, erythematous rashes, papular rashes, generalized rash, maculopapular rash; very rarely - alopecia.
From the musculoskeletal system:
often - back pain, arthralgia, pain in the limbs, in elderly patients with dementia - gait disturbances, swelling of the joints, in children - myalgia, neck pain; Uncommon: muscle pain in the chest, joint stiffness, muscle weakness, rhabdomyolysis.
From the urinary system:
often - urinary incontinence, in children - enuresis, pollakiuria; uncommon - pain when urinating; very rarely - urinary retention.
From the reproductive system:
often - lack of ejaculation, in children - galactorrhea; uncommon - menstrual irregularities, amenorrhea, gynecomastia, vaginal discharge, erectile dysfunction, ejaculation disorders, breast enlargement, sexual dysfunction, retrograde ejaculation; very rarely - priapism.
From the side of metabolism:
often - fatigue, asthenia, fever, pain in the chest; in elderly patients with dementia - peripheral edema, gait disturbance; in children - slowness, discomfort in the chest; infrequently - thirst, flu-like condition, edema, poor health, facial swelling, general edema, chills, cold extremities, withdrawal syndrome; very rarely - hypothermia.
From the laboratory parameters:
often - increased CK activity, increased heart rate, in elderly patients with dementia - increased body temperature, in children - increased body weight, increased prolactin concentration; uncommon - increased ALT activity, ECG disturbance, increased number of eosinophils in the blood, increased AST activity, increased number of leukocytes in the blood, increased blood glucose levels, decreased hemoglobin concentration, decreased hematocrit, decreased body temperature, decreased blood pressure; very rarely - prolongation of the QT interval on the ECG.
Below are additionally listed the adverse reactions that are observed when using risperidone intramuscularly (the drug Rispolept Konsta).
Infections:
lower respiratory tract infections, gastroenteritis, subcutaneous abscess.
From the blood and lymphatic system:
neutropenia.
From the side of metabolism:
hyperglycemia, weight loss.
From the mental side:
depression, sleep disorders.
From the nervous system:
paresthesia, convulsions.
From the senses:
blepharospasm, vertigo.
From the cardiovascular system:
bradycardia, increased blood pressure.
From the digestive system:
toothache, tongue spasm, increased GGT activity.
Dermatological reactions:
eczema.
From the musculoskeletal system:
pain in the buttocks.
From the reproductive system:
delayed menstruation, delayed ejaculation, oligomenorrhea, discomfort in the mammary glands.
Other:
pain, gait disturbances, detection of glucose in the urine, increased activity of microsomal liver enzymes.
Rispolept, 20 pcs., 2 mg, film-coated tablets
Inside,
regardless of food intake.
Schizophrenia
Adults.
Rispolept® can be prescribed 1 or 2 times a day. The initial dose of Rispolept® is 2 mg/day. On the second day, the dose should be increased to 4 mg/day. From this point on, the dose can either be kept at the same level or individually adjusted if necessary. Typically the optimal dose is 4–6 mg/day. In some cases, a slower dose escalation and lower initial and maintenance doses may be justified.
Doses above 10 mg/day do not show greater efficacy than lower doses and may cause extrapyramidal symptoms. Due to the fact that the safety of doses above 16 mg/day has not been studied, doses above this level cannot be used.
Benzodiazepines can be added to Rispolept® therapy if additional sedation is required.
Elderly patients.
The recommended initial dose is 0.5 mg per dose 2 times a day. The dose can be individually increased by 0.5 mg 2 times a day to 1–2 mg 2 times a day. If it is necessary to use a dose of 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Teenagers over 13 years old.
The recommended initial dose is 0.5 mg once daily in the morning or evening. If necessary, the dose can be increased after at least 24 hours by 0.5–1 mg per day to the recommended dose of 3 mg/day if well tolerated. The safety of doses above 6 mg/day has not been studied.
For patients who experience persistent drowsiness, it is recommended to take half the daily dose 2 times a day.
If it is necessary to use a dose of 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
There is no information on the use of the drug for the treatment of schizophrenia in children under 13 years of age.
Behavioral disorders in patients with dementia
The recommended initial dose is 0.25 mg per dose 2 times a day. If necessary, the dose can be increased individually by 0.25 mg 2 times a day, no more than every other day. For most patients, the optimal dose is 0.5 mg 2 times a day. However, some patients are advised to take 1 mg 2 times a day.
Once the optimal dose is reached, it may be recommended to take the drug once a day.
If it is necessary to use doses of 0.25 or 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Bipolar disorders with mania
Adults.
The recommended initial dose of the drug is 2 or 3 mg/day at a time. If necessary, this dose can be increased at least after 24 hours by 1 mg/day. For most patients, the optimal dose is 1–6 mg/day.
Teenagers and children over 10 years old.
The recommended initial dose is 0.5 mg once daily in the morning or evening. If necessary, the dose can be increased after at least 24 hours by 0.5–1 mg/day to the recommended dose of 2.5 mg/day if well tolerated. For most patients, the optimal dose is 0.5–6 mg/day.
If it is necessary to use a dose of 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
The safety of doses above 6 mg/day has not been studied.
For patients who experience persistent drowsiness, it is recommended to take half the daily dose 2 times a day.
There is no information on the use of the drug for the treatment of bipolar disorders in children under 10 years of age.
Behavioral disorders
Patients weighing 50 kg or more.
The recommended initial dose of the drug is 0.5 mg once a day. If necessary, this dose can be increased by 0.5 mg/day no more than every other day. For most patients, the optimal dose is 1 mg/day. However, for some patients, 0.5 mg/day is preferable, while some require an increase in dose to 1.5 mg/day. If it is necessary to use a dose of 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Patients weighing less than 50 kg.
The recommended initial dose of the drug is 0.25 mg once a day. If necessary, this dose can be increased by 0.25 mg/day no more than every other day. For most patients, the optimal dose is 0.5 mg/day. However, for some patients, 0.25 mg/day is preferable, while some require an increase in dose to 0.75 mg/day. If it is necessary to use doses of 0.25 or 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Long-term use of the drug Rispolept® in adolescents should be carried out under the constant supervision of a physician.
Use in children under 5 years of age has not been studied.
Autism in children and adolescents
The dose of Rispolept® should be selected individually. The recommended initial dose of the drug is 0.25 mg/day for patients weighing less than 20 kg and 0.5 mg/day for patients weighing 20 kg or more. On the 4th day of administration, the dose can be increased by 0.25 mg/day for patients weighing less than 20 kg and by 0.5 mg/day per day for patients weighing 20 kg or more.
This dose should be used until approximately the 14th day of treatment, when it is necessary to evaluate the effectiveness. Further increase in dose is carried out only in case of lack of effectiveness. The dose can be increased at intervals of 2 weeks or more by 0.25 mg/day for patients weighing less than 20 kg and by 0.5 mg/day for patients weighing 20 kg or more.
In clinical studies, the maximum daily dose did not exceed 1.5 mg/day for patients weighing less than 20 kg and 2.5 mg/day for patients weighing 20 kg or more, and 3.5 mg/day for patients with weight body more than 45 kg.
Table
Doses of the drug Rispolept® in the treatment of autism in children (per day)
| Body weight, kg | Days 1–3, mg | Days 4–14+, mg | Increase the dose (if necessary) | Recommended dose, mg |
| Less than 20 | 0,25 | 0,5 | +0.25 mg after 2 weeks or more | 0,5–1,5 |
| 20 or more | 0,5 | 1 | +0.5 mg after 2 weeks or more | 1–2,5* |
* For patients weighing more than 45 kg, higher doses may be required, the maximum dose studied is 3.5 mg/day.
If it is necessary to use doses of 0.25 or 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Rispolept® can be prescribed 1 or 2 times a day.
For patients who experience persistent drowsiness, it is recommended to take a daily dose 1 time before bedtime or 2 times a day.
If consistent efficacy is observed, the decision may be made to gradually reduce the dose to achieve an optimal balance of efficacy and safety.
Use in children under 5 years of age has not been studied.
Other patient groups
Patients with liver and kidney diseases.
Patients with kidney disease have a reduced ability to eliminate the active antipsychotic fraction compared to other patients. In patients with liver disease, there is an increased concentration of the free fraction of risperidone in the blood plasma.
The initial and maintenance dose, in accordance with the indications, should be reduced by 2 times; the dose increase in patients with liver and kidney diseases should be carried out more slowly.
If it is necessary to use doses of 0.25 or 0.5 mg, it is recommended to prescribe the drug Rispolept®, an oral solution.
Rispolept® should be prescribed with caution in this category of patients.